Description
USAGE AND SERVING SIZE
HOW SUPPLIED
1 FL OZ. (30 ML) Bottles
STORAGE
Store at room temperature in a dry place away from direct sunlight.
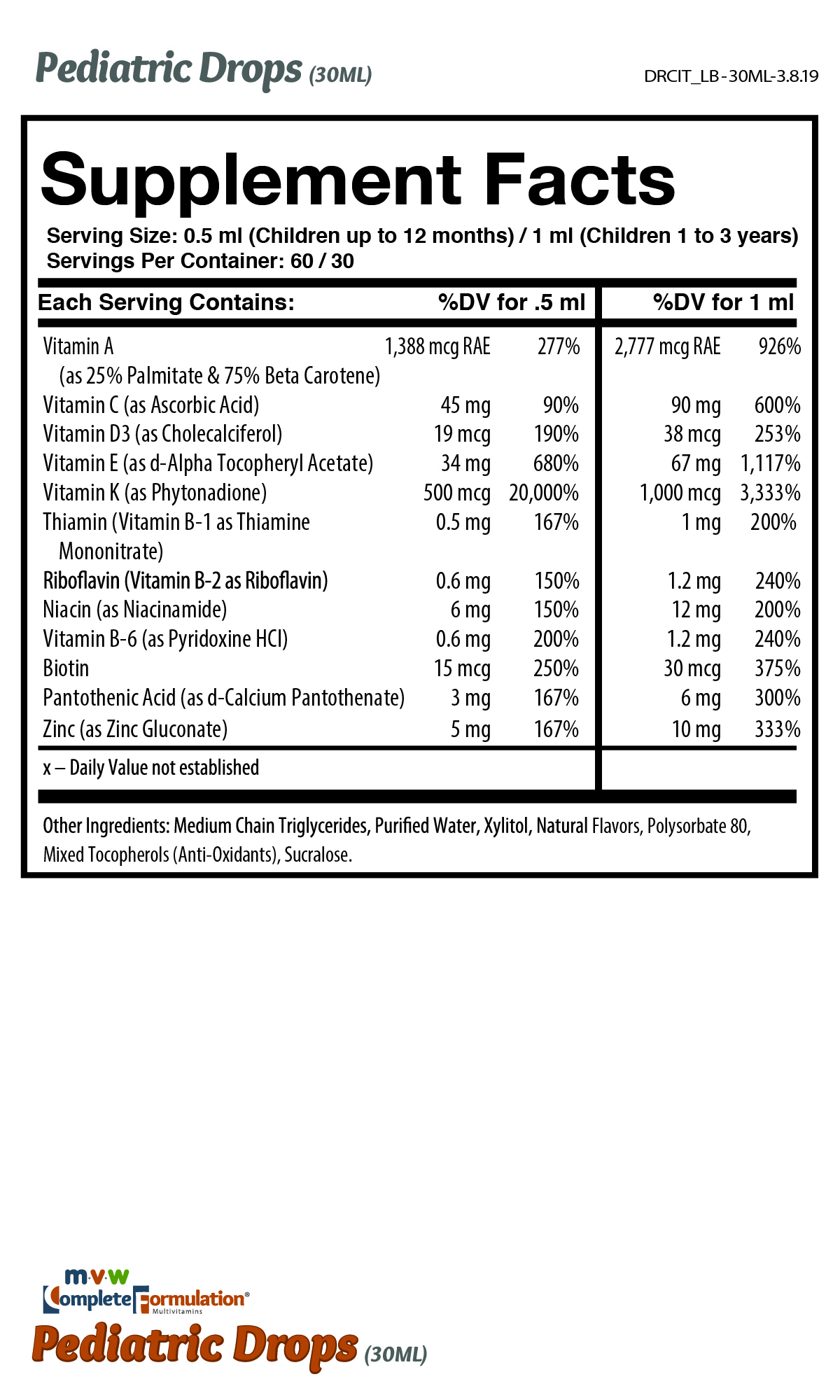
Serving Size
Children up to 12 months: 0.5 ml daily
Children 1 to 3 years: 1ml daily or as directed by a physician.
Shake contents before dispensing. To be taken with meals containing fat.
USAGE INFORMATION
MVW Complete Formulation Pediatric Drops may be used for nutritional supplementation in individuals whose diets are deficient in the fat-soluble vitamins A, D, E, and K.
For use solely under medical supervision.
These statement has not been evaluated by the Food & Drug Administration. This product is not intended to diagnose, treat, cure or prevent any disease.
SAFETY and PRECAUTIONS
MVW Complete Formulation Pediatric Drops is safety sealed for your protection. If the safety seal is broken or missing, do not use and contact MVW Nutritionals immediately at 1-855-236-8584.
Keep out of reach of children. Use caution when administering to children. In case of accidental overdose, call a doctor or poison control center immediately.
Supplement Facts
Same formulation. Any change in the amounts reflected on the label are due to new FDA values.
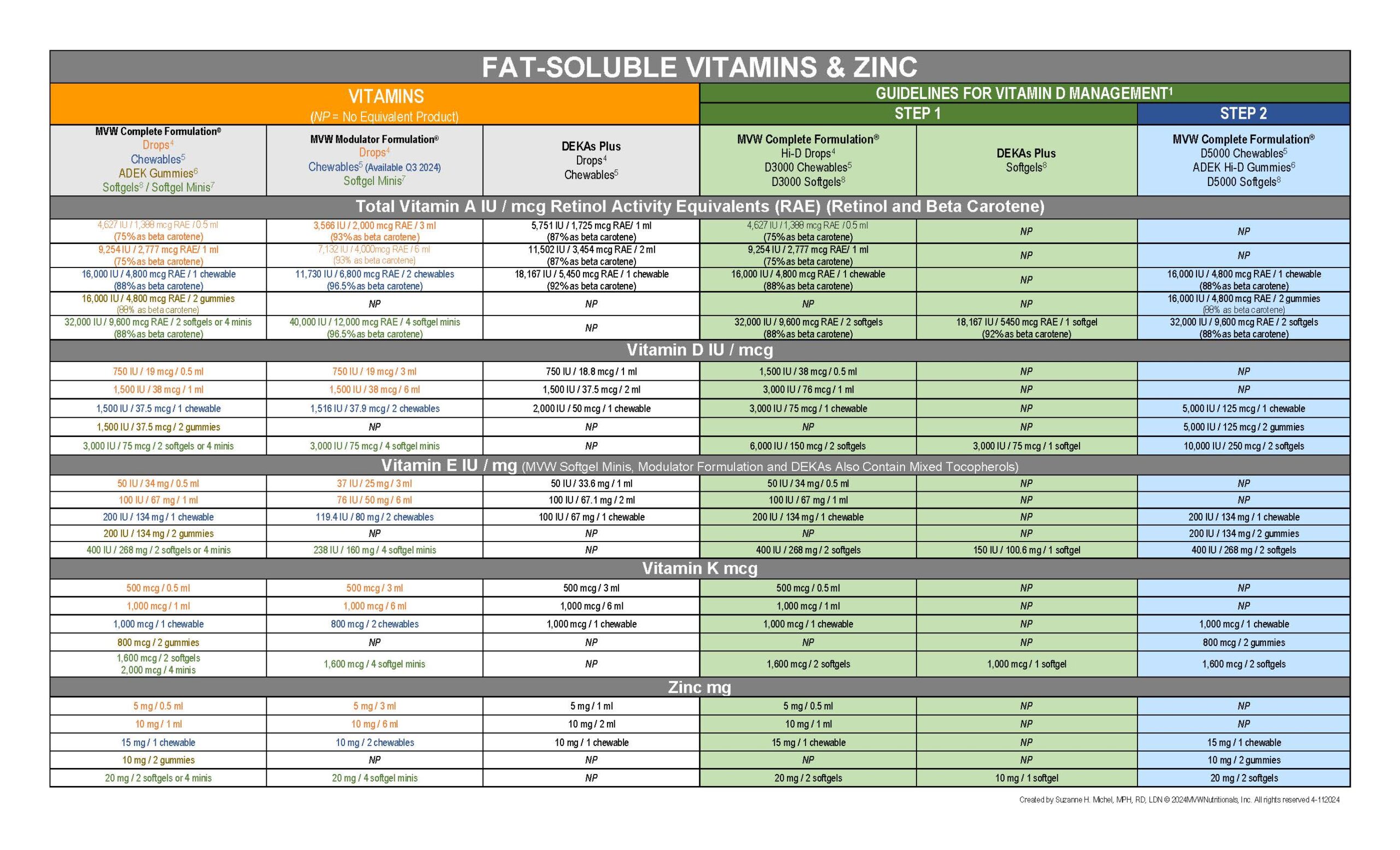
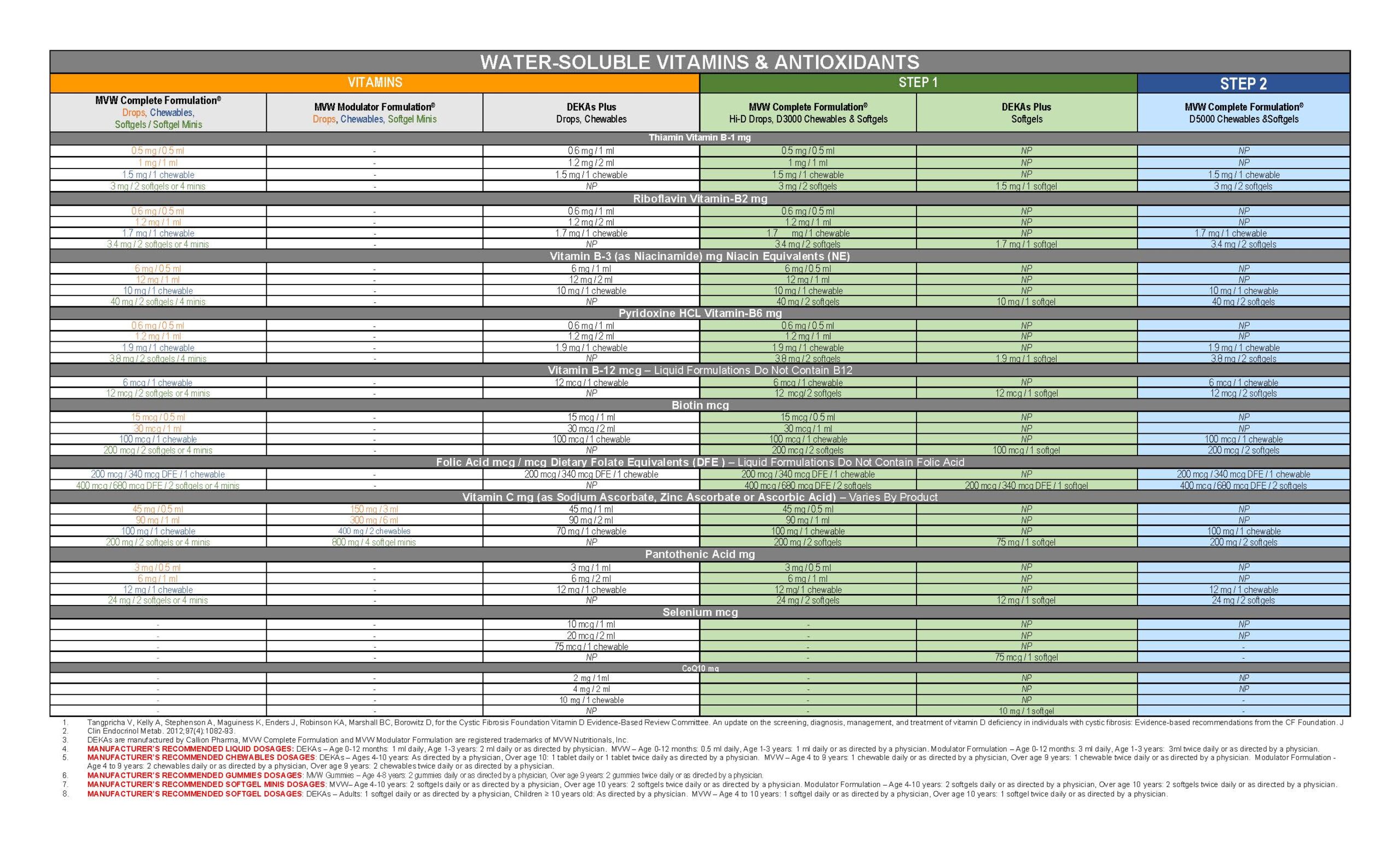
Vitamin Comparison Chart
MVW Complete Formulation Pediatric Drops are a lower dose, non-aqueous multivitamin supplement developed for people whose diets are deficient in the fat-soluble vitamins A, D, E, and K or who have difficulty absorbing fat-soluble vitamins.
MVW Complete Formulation Pediatric Drops provide the fat-soluble vitamins A, D, E, and K, plus B vitamins, vitamin C, and zinc. The pediatric drops also contain the antioxidants beta-carotene, ascorbic acid, alpha-tocopherol, mixed tocopherols, and zinc.
MVW Complete Formulation Drops have a natural citrus flavor.
MVW Complete Formulation Drops utilize a MCT oil based system to support absorption, as MCT oil is passively absorbed and is tasteless.
MVW Complete Formulation Drops are provided in 60ml/2oz bottles. Each bottle, however, due to the reduced serving size contain 30ml/1oz. To enhance stability, air that is commonly found in the headspace – the area between the liquid surface and top of container – is replaced with nitrogen, a stable, inert gas. Air, in addition to light and higher temperatures, are associated with a loss of potency in liquid vitamin formulations.
These statements has not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure or prevent any disease.
Are the MVW vitamins available through any special programs?
Yes, they are available through the AbbVie Care Forward program
(https://www.creon.com/CFPatients/CFCareForward)
and the Live2Thrive program
(https://www.live2thrive.org).
Ask about other program availability.
Are they available through my Specialty Pharmacy?
Our complete line of products are stocked by most of the Specialty Pharmacies across the country.
How are local pharmacists getting MVW Complete Formulation products through their wholesalers?
The Order Entry Numbers for MVW Nutritional Multivitamins – chewables, standard softgels, chewables and softgels with higher D, and drops are reflected in the chart below:
- Advise the pharmacist that the three major wholesalers – McKesson, ABC and Cardinal – have assigned order entry numbers for products that may be ordered through their respective wholesale distribution centers.
- The pharmacist should enter the number in their order entry system. If their local distribution center has already stocked the desired product, the order will process and product will arrive at to the pharmacist with the next shipment from the wholesaler – normally one to two days.
- If the local distribution center has never stocked the product, the order entry system will display one of two error messages: product discontinued, manufacturer backorder. Should the pharmacist get either of these two messages, they must CALL their local distribution center, provide the item number, and request that they stock the product. Distributors call this “activating an item” or “bringing the item in”. NOTE: Only an item with an assigned order entry can be activated at the local distribution center level in this manner.
- For products without an item number, the local distribution center must call their corporate office, and request that corporate assign an item number.
- If the pharmacist is (i) less than co-operative, or (ii) has questions, please call our toll-free number – 855-236-8584 – and we be happy to assist in this process.



Reviews
There are no reviews yet.